EPR on molecules and crystals
There is one essential difference between free atoms at one hand, and molecules or crystalline solids at the other hand: the spherical symmetry of the former. You will understand how spatial anisotropy leads to direction-sensitive EPR-measurements, and you’ll get to know how this can be used to identify, for instance, defects in solids.
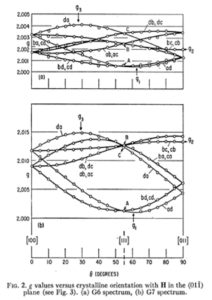
There are two tasks related to this video. The first one is meant to consolidate your understanding of the type of experimental EPR pictures which you saw in the video, and of which an example is repeated hereunder. Describe step by step, in an operational way, what you should do in an experiment to obtain a picture like this. Add your description to the forum.
- This forum has 16 topics, and was last updated 2 months ago by
Guillaume Smet.
-
- Topic
- Voices
- Posts
- Last Post
-
-
EPR
Started by:
Guillaume Smet
- 1
- 1
- 2 months ago
-
Answer
Started by:
Yaxi Li
- 1
- 1
- 2 months ago
-
Epr
Started by:
DMauricio34
- 1
- 1
- 2 months ago
-
anwer
Started by:
sennedebruyn
- 1
- 1
- 2 months, 1 week ago
-
answer
Started by:
nvrafelg
- 1
- 1
- 2 months, 1 week ago
-
answer1
Started by:
fedra_moeykens
- 1
- 1
- 2 months, 3 weeks ago
-
Recording data
Started by:
Hannes G.
- 1
- 1
- 1 year, 1 month ago
-
EPR on molecules and crystals
Started by:
Daniel
- 1
- 1
- 1 year, 1 month ago
-
EPR experiment
Started by:
WannesB
- 1
- 1
- 1 year, 2 months ago
-
EPR
Started by:
Niels Houben
- 1
- 1
- 1 year, 2 months ago
-
EPR multiple orientations
Started by:
ajsheffler
- 1
- 1
- 1 year, 2 months ago
-
EPR
Started by:
DanteBouckhout
- 1
- 1
- 1 year, 2 months ago
-
EPR experiment
Started by:
fdwit
- 1
- 1
- 1 year, 3 months ago
-
Experimental EPR pictures
Started by:
Jakob Vermeulen
- 1
- 1
- 1 year, 3 months ago
-
Experimental EPR pictures
Started by:
Iulia ZAI
- 1
- 1
- 2 years, 1 month ago
-
EPR
-
- You must be logged in to create new topics.
The second task aims to bring you in contact with more extensive EPR information (remember this in case you want to dig deeper, now or later). Please visit the website of this EPR biochemistry group at Auburn University. In particular, open their pdf-document called “short-short introduction to EPR spectroscopy“. It’s 42 pages, not that short after all. No urge to read it completely. However, start reading at page 1. You will recognize many concepts you have met before in hyperfinecourse. Keep on reading until you meet the first sentence you do not understand. Write down in the forum underneath where this sentence is in the document, and explain what you do not understand about it. Inspect the posts of other students in the forum, in particular the ones that got stuck at a sentence earlier in the document then you did — try to answer their question. That’s it for this task (but of course feel free to keep on reading in the pdf if you feel like).
- This forum has 13 topics, and was last updated 2 months ago by
Guillaume Smet.
-
- Topic
- Voices
- Posts
- Last Post
-
-
paper
Started by:
DMauricio34
- 1
- 1
- 2 months ago
-
paper
Started by:
sennedebruyn
- 1
- 1
- 2 months, 1 week ago
-
paper
Started by:
nvrafelg
- 1
- 1
- 2 months, 1 week ago
-
paper
Started by:
fedra_moeykens
- 1
- 1
- 2 months, 3 weeks ago
-
Paper
Started by:
Hannes G.
- 1
- 1
- 1 year, 1 month ago
-
paper
Started by:
Yannick
- 1
- 1
- 1 year, 1 month ago
-
Short-Short introduction.
Started by:
WannesB
- 1
- 1
- 1 year, 2 months ago
-
EPR introduction
Started by:
Niels Houben
- 1
- 1
- 1 year, 2 months ago
-
EPR Short-short introduction
Started by:
ajsheffler
- 1
- 1
- 1 year, 2 months ago
-
Paper
Started by:
DanteBouckhout
- 1
- 1
- 1 year, 2 months ago
-
paper
-
- You must be logged in to create new topics.
Expected time: 1h00 minutes (report)
B10-03